Wave Particle
Duality
Joseph Courtney
AP Physics C- Mr. D.A.
Young
Updated Friday, May 26, 2006
Wave Particle Duality is
just one more phenomenon that shows the non-intuitive-ness of reality.
The entire debate began between Christian Huygens an Isaac Newton. Huygens proposed a wave theory of light which
had many problems when attempting to describe many of the often observed
properties of light. Isaac Newton then
proposed his theory of light explaining that it was composed of corpuscles of
light. This allowed him to explain
reflection, refraction, and the splitting of light into a rainbow.
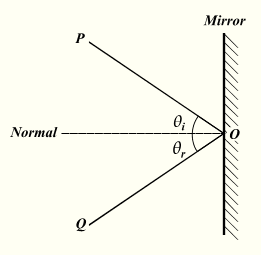
As

Then Einstein came
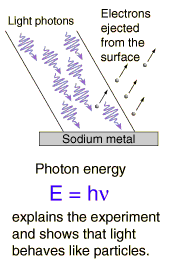
along. He studied the Photo-electric
effect where the excitation of electrons within a metal by light only occurs
when the light’s frequency is above a certain value. This implies that the energy of a photon is
related to the frequency of the photon rather than to the amplitude of the
“light wave” or to the intensity of the light.
Einstein then described this phenomenon by stating that the energy must
occur in packets of light, each having a certain frequency. He called these packets of light
photons. He also described the energy of
a photon by stating that its energy was described by the equation![]() where E is energy, h is Planck’s constant, and
where E is energy, h is Planck’s constant, and ![]() is the frequency of
the light. This caused an uproar and thoroughly confused people as to whether light
occurred in particles or waves.
is the frequency of
the light. This caused an uproar and thoroughly confused people as to whether light
occurred in particles or waves.

Then there was de
Broglie. He generalized Einstein’s
equation for the energy of light by taking the description of the momentum of
the photon, p=E/c, and the definition of the wave length of light as λ
= c / ν. He combined these to make his own equation λ
= h / p. This was generalized for all matter and
light. He used this equation to describe
the wave length of the electron which he then used to describe the quantized
nature of the electron energy levels of the hydrogen atom. He proposed that energy levels only occur
when the electrons orbit is a whole number of electron wavelengths which would
make the energy of each level also whole number intervals.
This
allows me to find my wavelength. If I were
driving down college at 20 meters per second and me being roughly 70kg on a
good day, I would have a momentum, p, of 1400 kg·m/s. Using the de Broglie equation, my wavelength
would be Planck’s Constant devided
by my momentum. This would mean that my wavelength
at that velocity is about 2.732x10^-37 m.
Since
the Particle-Wave, Wave-Particle Duality of matter and light has been accepted,
although not intuitively understood, by the majority of physicists. Many experiments have been performed to
attempt to diffract larger particles through a double-slit such as neutrons,
protons, water, and even Buckminster fullerene.
Theoretically a particle such as a bacterium would be impossible to
diffract due to its wavelength being smaller than the Planck length, the
theoretical quantization of the spatial dimensions. However, work is being done to attempt to
diffract virus particles.
Now
Wave-Particle Duality has been used to describe many natural phenomena such as
the interference caused by the fluctuating densities and thicknesses of soap
bubbles which give them their iridescent look, the polarization of light, the
reflection of light by Mirrors, classical optics, rainbows, why the sky is
blue, the many phenomena in chemistry, and why we can see.
I
acquired much of my information from these two websites
http://en.wikipedia.org/wiki/Wave-Particle_duality
http://hyperphysics.phy-astr.gsu.edu/hbase/mod1.html
FERROFLUIDS!
Other Cool Links:
http://hyperphysics.phy-astr.gsu.edu
Math, Physics, and Engineering
Applets
I hereby promise that I have
neither given nor received any unauthorized assistance on this here assignment
My name is Joseph Courtney
and I am a Physics Student at